 The general assumption of setting up control charts and QC rules is that you are selecting rejection rules. You stop the process, determine and fix what's wrong, then resume production. The basic idea of the control chart is that if a control rule is violated, your system is out-of-control. Here's a short list of interpretation styles: Over the years, we have seen laboratories interpret the out-of-control flag in many ways. While it isn't complex to figure out whether or not the data has violated a particular control rule, what to do after that is often hard to figure out. Well, the reality is not quite as simple. Throw some lines on a chart, and if the dot is is above the line, it's out, right? As other lessons on the website detail, the definitions of the control rules are fairly straightforward, but the selection of which rules to use for a given test is more complicated. Defining statistical control rules should be simple enough. 
When it comes to quality control in the medical laboratory, it seems that the complexity hides in every step. What's the Role of a Rule? Sten Westgard, MS 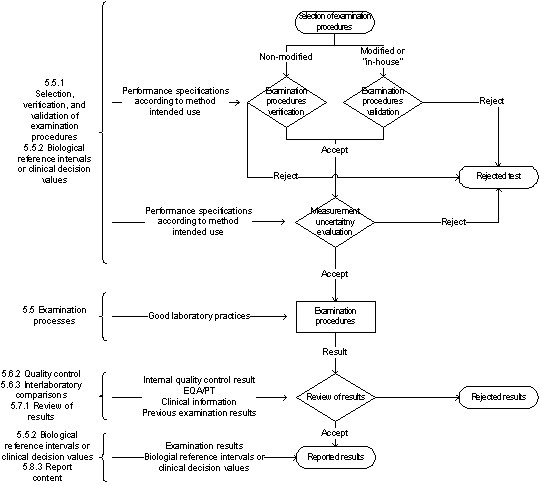
This article is exclusively sponsored by LGC Technopath Clinical Diagnostics Now that you've got your control chart set up, your controls running, and your data plotting, what do you do with the rules when the dots are out? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
